XB-IMG-128132
Xenbase Image ID: 128132
|
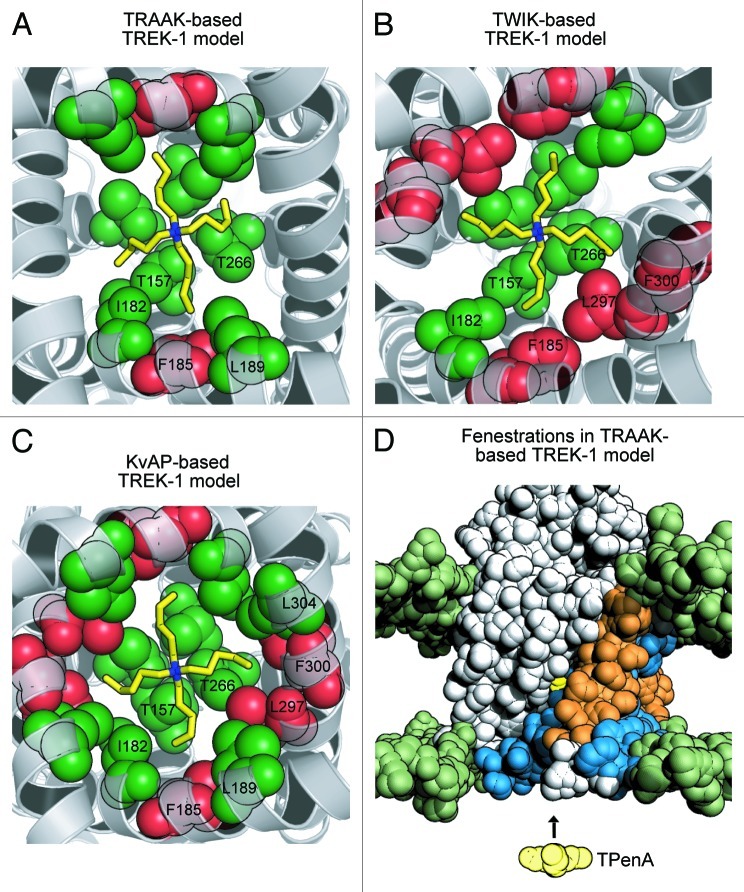
Figure 1. Refined structural models of TREK-1 with TPenA bound. (A) Bottom up view of a structural model of the TREK-1 pore with docked TPenA. The model was based upon the crystal structure of TRAAK (PDB ID: 3UM7). Those residues which interact with TPenA are highlighted as spheres and colored green if they agree with our functional scanning mutagenesis study or red if a false positive. Only residues on one protomer are numbered. (B) Similar model based upon the crystal structure of TWIK-1 (PDB ID: 3UKM). (C) Similar model based upon the crystal structure of KvAP (PDB ID: 1ORQ). (D) Solvent-accessible surface area (SASA) of a TREK-1 model, based on TRAAK, in which the side-fenestrations are still clearly visible. The SASA of the phospholipid head groups is colored green whereas the TREK-1 model is colored gray. For one protomer, the outer (M1 and M3) and inner (M2 and M4) transmembrane helices are highlighted in orange and blue, respectively. The docked TPenA is also visible through the side-fenestration (colored in yellow spheres) and QA ion access to this binding site occurs via the cytoplasmic pore of the channel (indicated by direction of the arrow). Image published in: Rapedius M et al. (2012) Copyright © 2012 Landes Bioscience. This image is reproduced with permission of the journal and the copyright holder. This is an open-access article distributed under the terms of the Creative Commons Attribution-NonCommercial license Larger Image Printer Friendly View |
