XB-IMG-130817
Xenbase Image ID: 130817
|
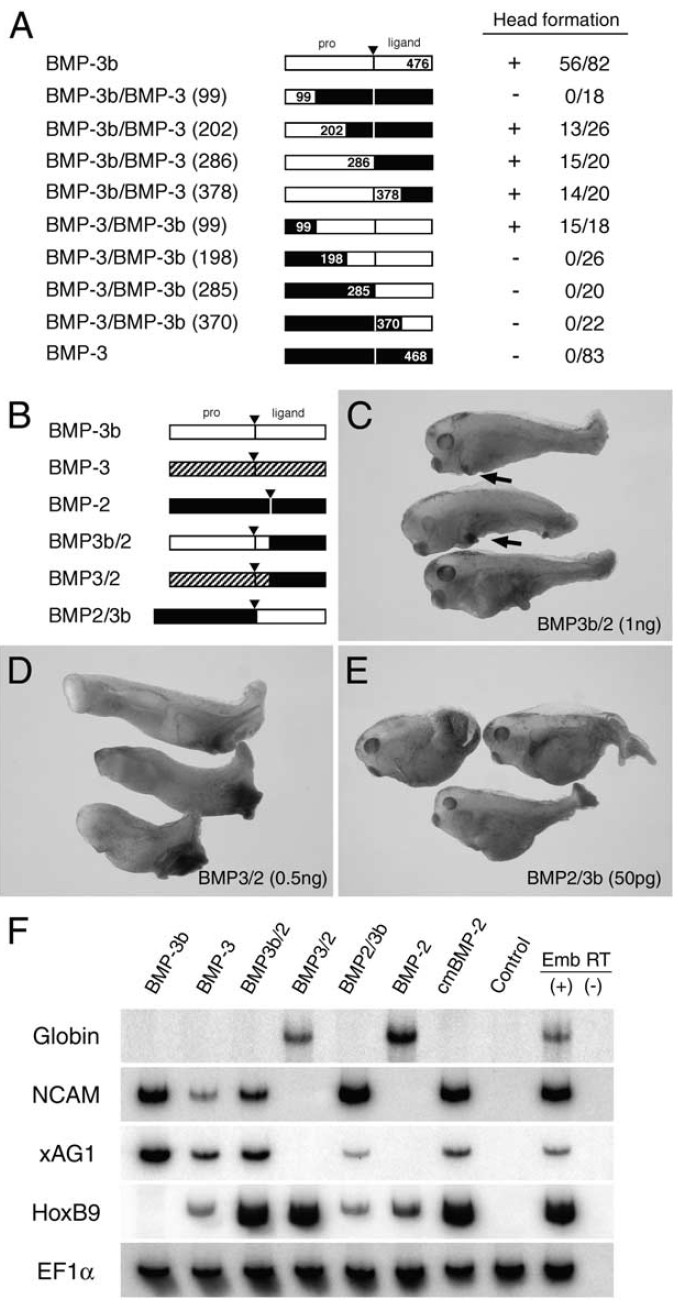
Fig. 5. Distinct activities of BMP-3b and BMP-3 are caused by differences in
their pro-domains. (A) Chimera analysis of BMP-3b and BMP-3 for localizing
BMP-3b head-forming activity. Pro-domain of BMP-3b protein is required for
head formation. Activity and score of head formation are tabulated on the right.
Numbers represent positions of recombination. (B) Schematic representation
of chimeras between either BMP-3b or BMP-3, and BMP-2. Pro-domains of
BMP-3b and BMP-3 were fused to ligand domain of BMP-2, and ligand
domain of BMP-3b was combined with BMP-2 pro-domain. Synthetic mRNAs
encoding these chimeras were injected into marginal zones of two ventral
blastomeres at the four cell stage. (C) Injecting BMP3b/2 chimera mRNA (1
ng) perturbed tail formation and induced cement gland (arrows) (96%, n
25). (D) Overexpression of BMP3/2 mRNA (500 pg) ventralized embryos
(100%, n 25). (E) BMP2/3b chimera (50 pg) generated same phenotype as
BMP-3 (100%, n 24). (F) Animal cap assay of chimera functions. Caps
injected with mRNAs (200 pg each) were isolated at stage 8.5, and harvested
at stage 23 to score by RT-PCR. BMP-3b induced NCAM and xAG1, whereas
BMP-3 induced these markers and HoxB9. BMP3b/2 and BMP2/3b chimeras
induced NCAM and xAG1, whereas BMP3/2 induced globin, suggesting that
pro-domain of BMP-3, but not of BMP-3b allows cleavage of ligand domain.
Moreover, ligand domains of BMP-3 and BMP-3b have activity antagonistic
to that of BMP-2. We also found that BMP-2 and its cleavage site mutant
(cmBMP-2) induced HoxB9, a posterior neural marker. This observation may
relate to a previous finding that HoxB9 is expressed weakly in lateral plate
mesoderm as well as in the spinal chord (Wright et al., 1990; see also Fig. 9J). Image published in: Hino J et al. (2003) Copyright © 2003. Image reproduced with permission of the Publisher, Elsevier B. V. Larger Image Printer Friendly View |
