XB-IMG-130819
Xenbase Image ID: 130819
|
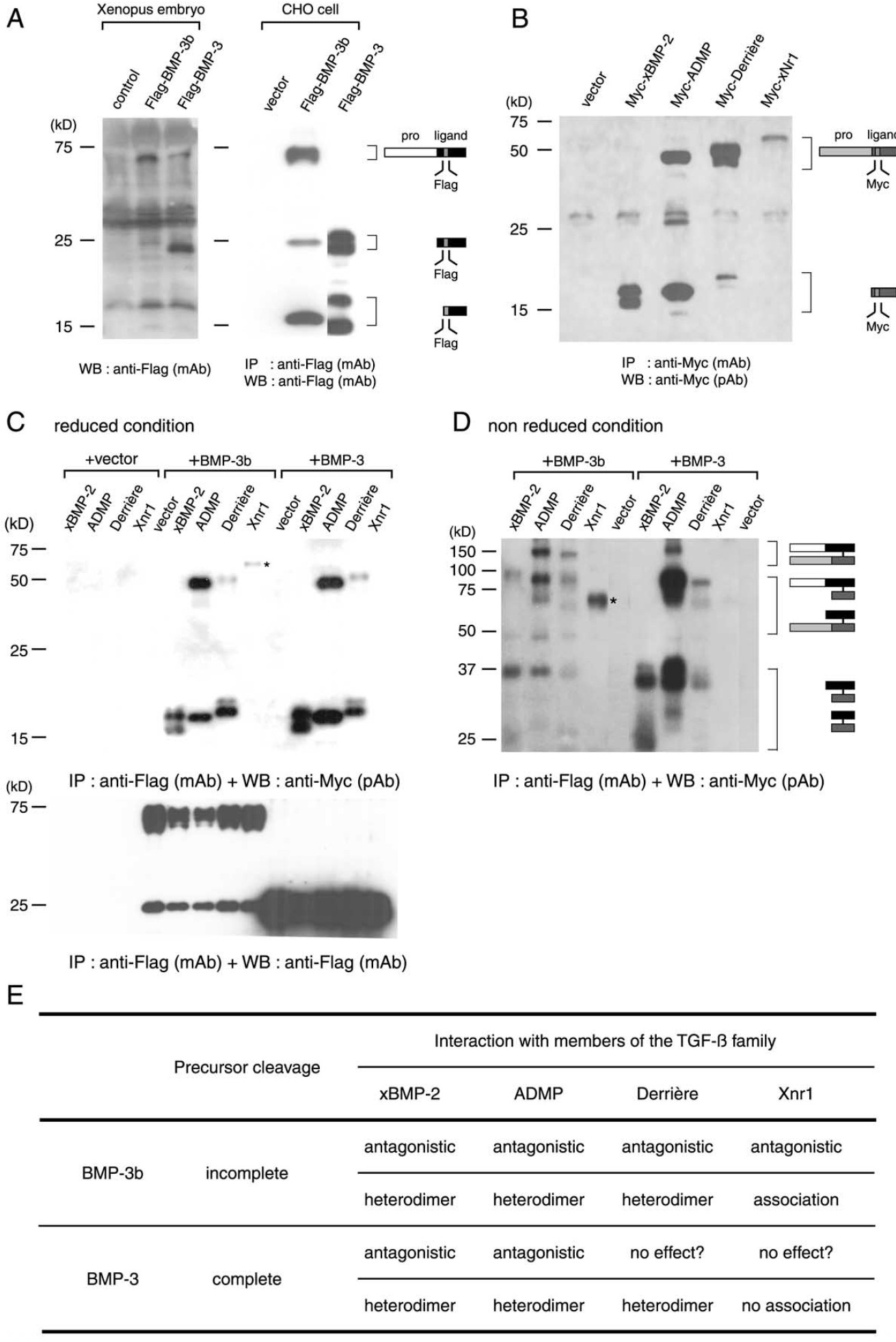
Fig. 7. Divergence of proteolytic processing and heterodimer formation of BMP-3b and BMP-3. (A) Less BMP-3b than BMP-3 precursor was cleaved in
Xenopus embryos and in CHO cells. Extracts of embryos injected with Flag-tagged BMP mRNAs (left panel) and conditioned media of CHO cells transfected
with the plasmid DNAs (right panel) were Western blotted. Controls were from wild type embryos and from CHO cells that had been transfected with vector
DNA, respectively. All samples were resolved under reducing conditions. Molecular mass (kDa) of proteins (left), deduced structures, and Flag epitope (right)
are shown. Two bands generated by N-glycosylation were grouped at about 25 and 16 kDa, respectively (lane: BMP-3 expressed in CHO cells). (B)
Myc-tagged proteins of the TGF- family were constructed and expressed in CHO cells to test interactions with BMP-3b and BMP-3. Although we
transfected cells with equal amounts of each plasmid, Xnr1 precursor was not effectively processed and secreted from CHO cells. (C, D) Immunoprecipitation
and Western blotting revealed that BMP-3b directly interacted with all TGF- members, whereas BMP-3 interacted with all except Xnr1, in CHO cells.
Coprecipitated BMP-3b and BMP-3 are shown at 75 and 25 kDa in (C). Apparent molecular weight of Xnr1 (*) under reducing and nonreducing conditions
did not change, suggesting that BMP-3b forms noncovalent complexes with Xnr1. (E) Summary of molecular characteristics of BMP-3b and BMP-3 including
results of Figs. 5 and 6. Both BMP-3b and BMP-3 oppose BMP-2 and ADMP. BMP-3b also blocks derrie`re and Xnr1. Types of BMP-3b antagonism are
instructed by the pro-domain, which controls cleavage and assembly of the ligand domain. Incomplete processing of BMP-3b might reduce mesoderminducing
activity of derrie`re and complex formation between BMP-3b and Xnr1 might result in altered Xnr1 function. Image published in: Hino J et al. (2003) Copyright © 2003. Image reproduced with permission of the Publisher, Elsevier B. V. Larger Image Printer Friendly View |
