XB-IMG-159427
Xenbase Image ID: 159427
|
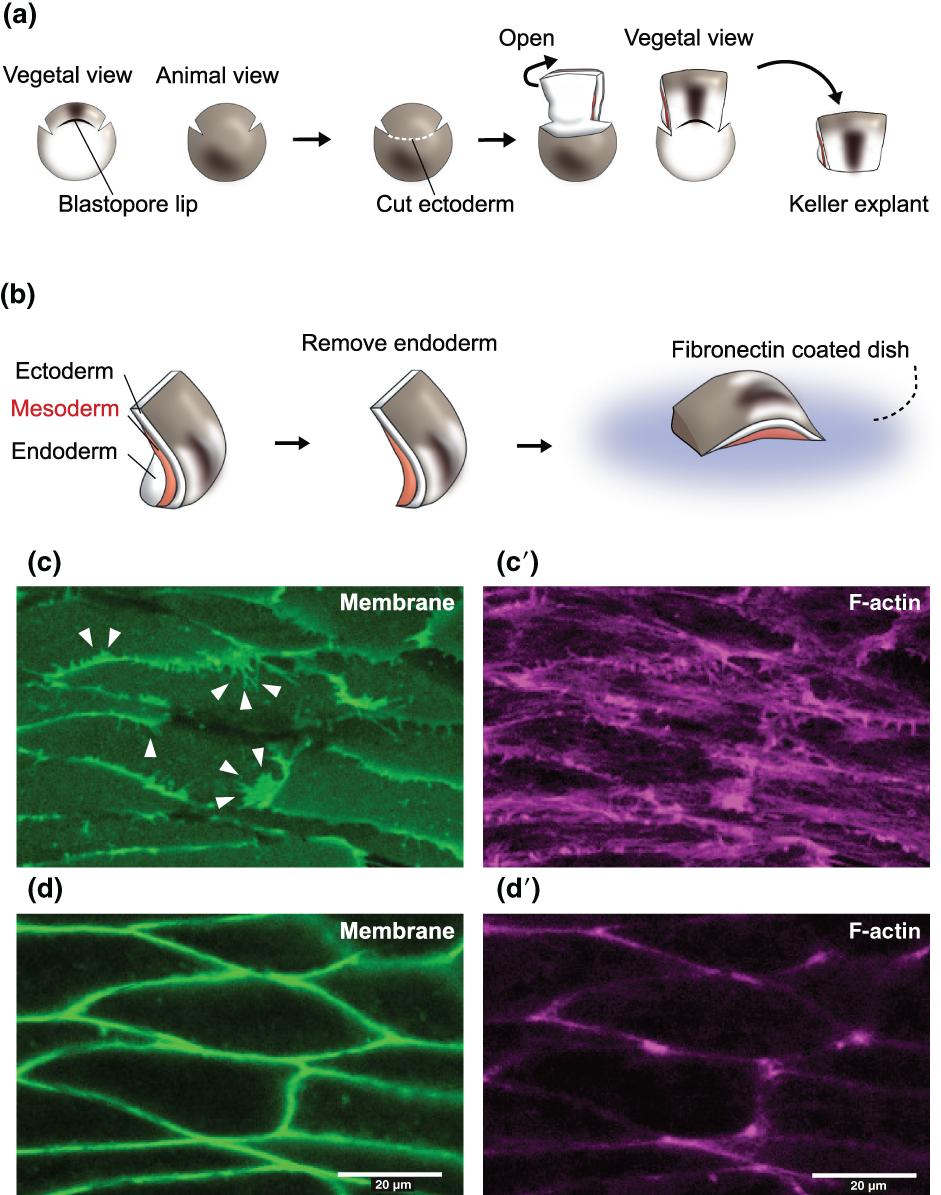
Figure 8. Tissue explant isolation from Xenopus laevis embryos for live imaging. (a) Procedure of isolating Keller explants. The explant is cut out at embryonic stage 10.5. Incisions are made on both sides of the blastopore lip, and the dorsal region is opened after cutting the ectoderm. The dorsal region is discerned by cutting along the blastopore lip. (b) Trimming the Keller explant and imaging the notochord. The endoderm is removed to expose the mesoderm (notochord) before mounting on a fibronectin-coated dish. The mesoderm is placed face down for the purpose of live imaging through inverted confocal microscopy. (c–d′) Images of the cell membrane and F-actin captured by live imaging of Keller explants. (c) and (c′) are images of the cells at the surface of the explant, where the cells adhere to the fibronectin. (d) and (d)′ are images of the same cells as in (c) and (c′), but focused 5 µm deep from the plane of (c) and (c′). The membrane protrusions (arrowheads) and actin cables are more obvious in (c) and (c′) images, whereas the borders of the cells are more obvious in (d) and (d’) images. Image published in: Shindo A (2018) © 2017 The Authors. Creative Commons Attribution-NonCommercial license Larger Image Printer Friendly View |
