XB-IMG-174946
Xenbase Image ID: 174946
|
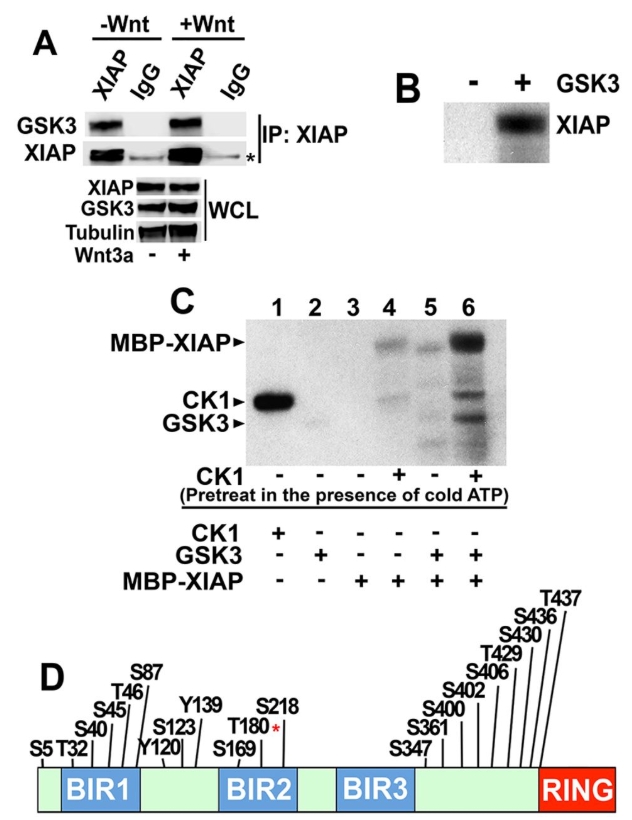
Fig. 1.
XIAP is phosphorylated by GSK3 at T180. (A) Endogenous XIAP co-immunoprecipitates with endogenous GSK3. XIAP was immunoprecipitated (IP) from whole-cell lysates (WCL) of HEK293STF cells incubated in the absence or presence of recombinant Wnt3a followed by immunoblotting. The asterisk indicates the heavy chain of IgG. (B) Purified XIAP is phosphorylated by GSK3 in vitro in a [γ32P]ATP kinase assay. Reactions were analyzed by SDS/PAGE followed by autoradiography. (C) CK1 enhances XIAP phosphorylation by GSK3. XIAP bound to beads was incubated with CK1 in a kinase reaction containing non-radioactive (cold) ATP (lanes 4 and 6). CK1 was washed away and XIAP-bound beads incubated with GSK3 in a kinase reaction containing [γ32P]ATP. Reactions were analyzed by SDS/PAGE followed by autoradiography. Kinase only lanes show CK1 and GSK3 autophosphorylation (lanes 1 and 2). Results in AâC were replicated at least three times. (D) The predicted phosphorylation site at T180 of XIAP is an in vitro GSK3 phosphorylated site. Predicted phosphorylation sites on XIAP were curated by the PhosphoSitePlus online resource tool (Hornbeck et al., 2015). LC-MS analysis of XIAP identifies T180 (asterisk) as a prominent in vitro GSK3 phosphorylation site. Image published in: Ng VH et al. (2018) Copyright © 2018. Image reproduced with permission of the Publisher, The Company of Biologists Ltd. Larger Image Printer Friendly View |
