2016 John Gurdon Prize Winner
16th International Xenopus Conference Presentation Award Winners
John Gurdon Trainee Talk - $500 Cash
Rachel Stephenson
University of Michigan
Email: restep@umich.edu
Post Doc – 1st Prize Winner $200 Amazon Vouchers
Helen Willsey
UC Berkeley/UCSF
Email: hrwillsey@gmail.com
Post Doc – 2nd Prize Winner £100 Amazon Vouchers
Eamon Dubaissi
University of Manchester
Email: Eamon.Dubaissi@manchester.ac.uk
Student/Phd – 1st Prize Winner $200 Amazon Vouchers
Grace Goh
A*STAR Institute of Medical Biology, Singapore
Email: grace.goh@imb.a-star.edu.sg
Student/Phd – 2nd Prize Winner £100 Amazon Vouchers
Rita Monteiro
Francis Crick Institute
Email: rita.monteiro@crick.ac.uk
The John Gurdon Award is given by the International Xenopus Board in recognition of the best talk from a trainee (student or postdoc) at the International Xenopus Conference. Inaugurated in 2014, this award honors Sir John Gurdon, whose Nobel Prize-winning work on pluripotency and nuclear reprogramming using Xenopus was initiated while he was a graduate student.
The inaugural prize (2014) was decided to be a tie, so the award went to Andrea Wills (Julie Baker Lab) and Yuuri Yasuoka (Masanori Taira lab).
Congratulations to Rachel Stephenson for winning the John Gurdon Prize for the best trainee presentation at the recent 16th International Xenopus Conference in Crete!

Rachel Stephenson (pictured on the right) is beginning her 5th year as a graduate student in Dr. Ann Miller’s Lab in the Molecular, Cellular, and Developmental Biology Department at the University of Michigan. The research in the Miller Lab investigates how Rho small GTPases regulate cytokinesis and cell-cell junctions in epithelial tissues. During development, tissue maintenance, and disease, epithelial tissues are frequently faced with events that disrupt epithelial homeostasis including cell intercalation, cell extrusion, and cytokinesis. The Miller Lab is studying how epithelial cells are able to maintain cell adhesion and barrier function while also remodeling their cell-cell junctions when faced with such challenges to epithelial homeostasis. It is known that precisely localized zones of active RhoA, a small GTPase that promotes actomyosin contractility, are required both for cytokinesis and for formation and maintenance of cell-cell junctions. However, little is known about how localized RhoA activity is regulated in space and time or how active RhoA and its downstream targets regulate junction maintenance, remodeling, and tension in dividing epithelial cells. To address these questions, high-resolution live confocal microscopy in the epithelium of Xenopus laevis embryos is their key approach. This allows the Miller Lab to learn about the molecular mechanisms that normally regulate cytokinesis and cell-cell junction remodeling and may lead to new insights into how defects in these processes may contribute to disease states including to tumor formation and metastasis.
At the 16th International Xenopus Conference, Rachel presented work demonstrating that Rho flares (localized bursts of RhoA activity at cell-cell junctions) function to rapidly repair tight junctions. Rho flares were first described in gastrula-stage Xenopus laevis embryos by the Miller lab (Reyes et al., 2014), and Rachel’s thesis work is focused on understanding what triggers Rho flares and what their function is. Rachel found that Rho flares are preceded by local discontinuities in tight junction proteins that are reinforced following the Rho flare. In her talk, Rachel presented data showing that Rho flares likely reinforce tight junctions through two distinct mechanisms: junction contraction and actin polymerization. This work is important for understanding how epithelial tissues maintain a stringent barrier in the face of various cell and tissue scale forces that put stress on the junctions (e.g., cytokinesis and morphogenesis).
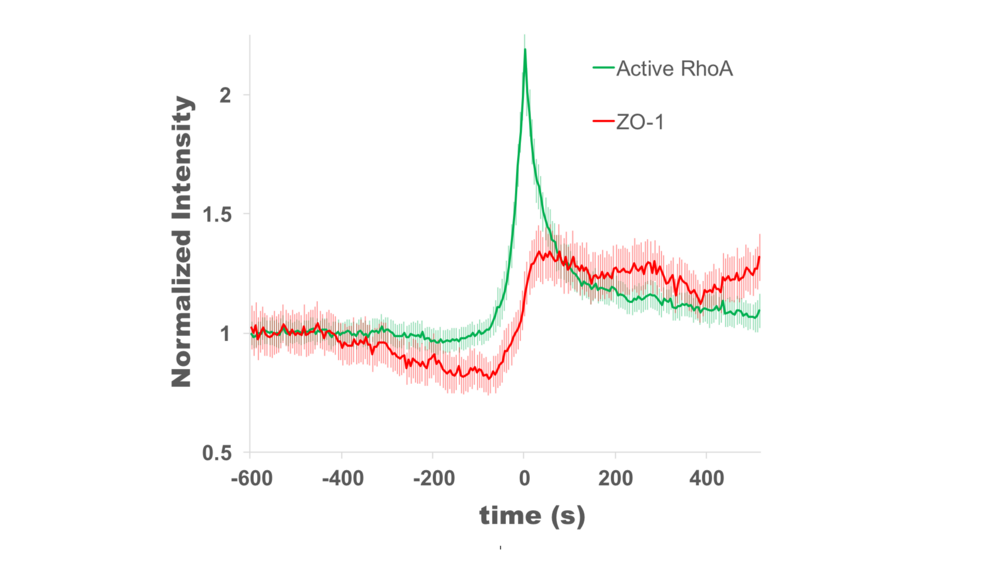
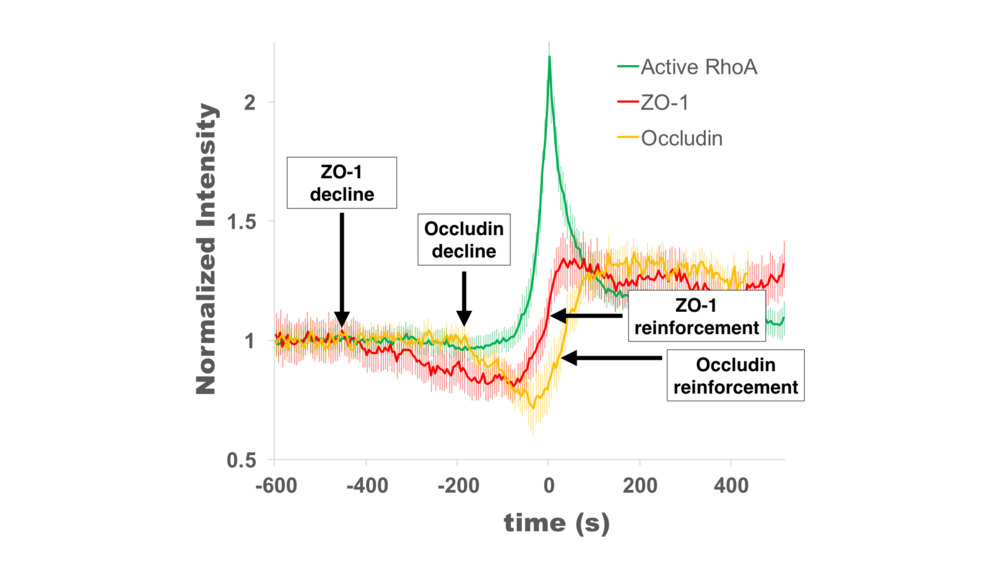

1. Reyes, C. C. et al. Anillin regulates cell-cell junction integrity by organizing junctional accumulation of Rho-GTP and actomyosin. Curr. Biol. 24, 1263–1270 (2014).
Congratulations to Helen Willsey for winning the 1st Place Post-doc Poster Prize at the recent 16th International Xenopus Conference in Crete!
Dr. Helen Willsey is a member of the laboratory of Dr. Richard Harland in the Department of Molecular and Cell Biology at the University of California, Berkeley.
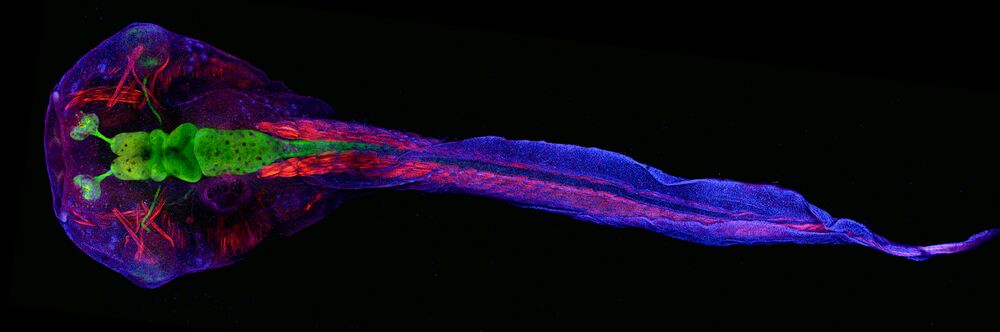
Research description: Autism spectrum disorder (ASD) is a devastating neurodevelopmental disorder of undetermined etiology, but recent advances in genomic approaches have led to the identification of over 65 ASD risk genes. We leverage Xenopus tropicalis and the CRISPR/Cas9 system to identify convergent phenotypes among ASD genes during neurodevelopment with the goal of understanding ASD pathology.
Congratulations to Eamon Dubaissi for winning the 2nd Place Post-doc Poster Prize at the recent 16th International Xenopus Conference in Crete!
Research description: His work focuses on the developing epidermis of the Xenopus tropicalis tadpole. The epidermis has both motile multi-ciliated cells and cells that secrete a mucin-like glycoprotein with the capacity to form a mucus barrier at its surface. In this way it closely resembles the human respiratory epithelia in the lungs. He is investigating the way in which the tadpole uses this barrier to protect itself against infection using the known opportunistic pathogen Aeromonas hydrophila. He finds that depletion of the major mucin-like glycoprotein from the skin leads to an increased sensitivity to infection with A. hydrophila.
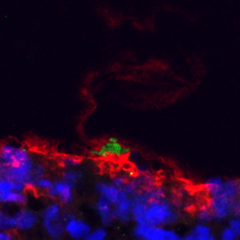
Snap frozen tadpole skin shows the mucin-like glycoprotein (red) trapping Aeromonas hydrophila (green) at the surface. DAPI in blue
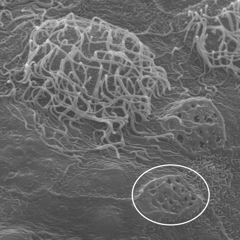
Scanning electron microscope image of the skin surface showing multi-ciliated cells and secretory cells (circled)
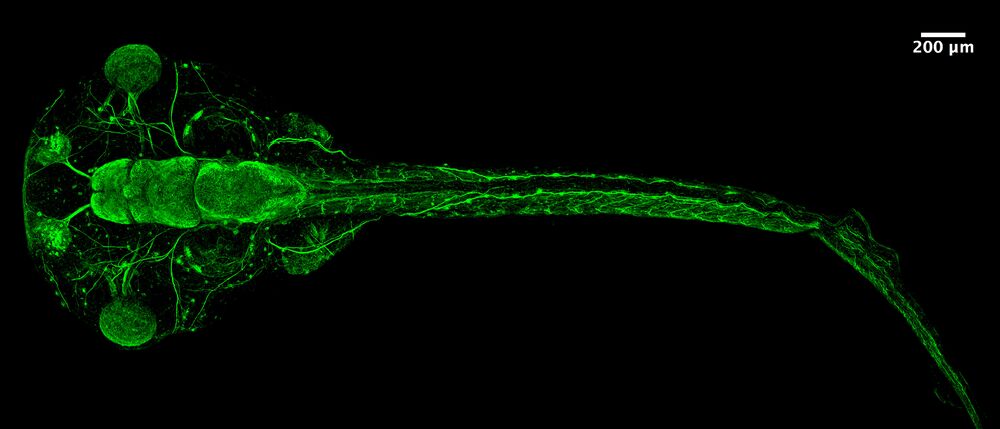
Congratulations to Grace Goh for winning the 1st Place Student/PhD Prize for her poster presentation at the recent 16th International Xenopus Conference in Crete!

The Reversade lab uses Xenopus, and other animal models, to investigate the mechanisms of genetic orphan diseases in humans. In the course of our studies, we discovered numerous small ORFs in the human genome that encode endogenous secreted peptides (e.g. www.elabela.com). Each of these putative hormones is being validated, de-orphanized and their possible clinical indication(s) evaluated.
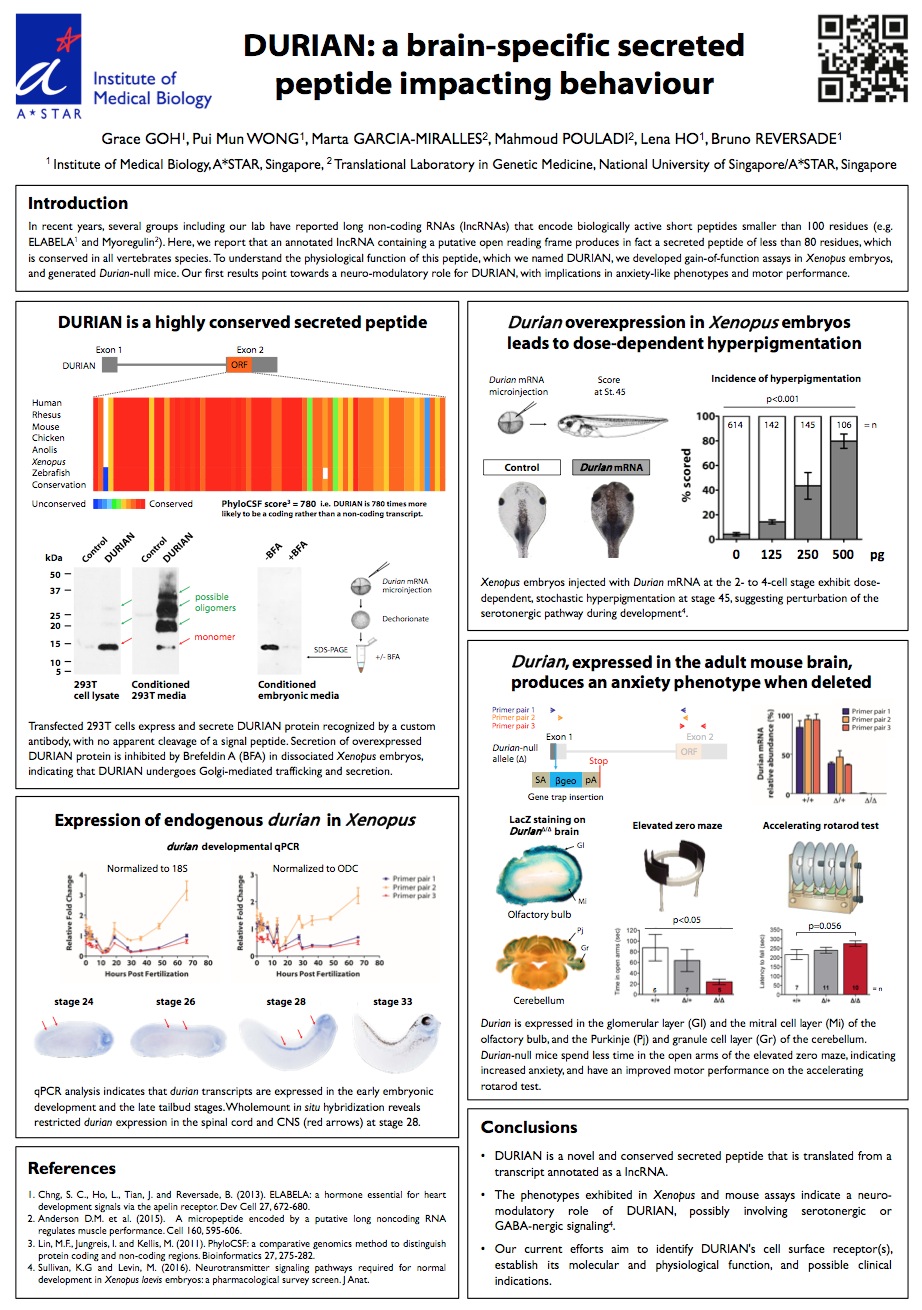
Click here to view the pdf of her poster.
Congratulations to Rita Montiero for winning the 2nd Place Student/PhD Prize for her poster presentation at the recent 16th International Xenopus Conference in Crete!
One of the main interests in Jim Smith’s Lab is mesoderm development and its regulatory players, such as the T-box transcription factor Brachyury. We have generated knockdown (KD, by splice- and translation-blocking Morpholinos) and knockout (KO, by TALEN-mediated mutagenesis) Xenopus embryos for Brachyury (t and t2). In an effort to understand the implications of Morpholinos for functional studies we compared Brachyury KD to KO embryos both phenotypically and transcriptionally. While both loss-of-function methods strongly affect the same core gene regulatory network leading to identical phenotypes (see figure), a small group of genes shows consistent mis-regulation only in morphants potentially causing off-target affects in later development.

Last Updated: 2016-10-10
