Digital dissection of Xenopus laevis using contrast-enhanced computed tomography
J Anat. August 1, 2017; 231 (2): 169-191.
Digital dissection of the model organism Xenopus laevis using contrast-enhanced computed tomography.
Porro LB , Richards CT.
Click here to view article at Journal of Anatomy.
Download article pdf.
Download interactive pdf documents: download and open in Acrobat reader recommended to enable interactivity.
joa12625-sup-0001-FigS1.pdf PDF document, 6139K
Fig. S1. Interactive 3D PDF of the digitally segmented skeleton of Xenopus laevis.
joa12625-sup-0002-FigS2.pdf PDF document, 10107K
Fig. S2. Interactive 3D PDF of the digitally segmented musculature of Xenopus laevis; with the exception of m. longissimus dorsi, only right side muscles are shown.
joa12625-sup-0003-FigS3.pdf PDF document, 7166K
Fig. S3. Interactive 3D PDF of the digitally segmented nervous system of Xenopus laevis.
joa12625-sup-0004-FigS4.pdf PDF document, 5231K
Fig. S4. Interactive 3D PDF of the digitally segmented digestive, urogenital and respiratory systems of Xenopus laevis.
Click here to view article on Pubmed.
Click here to view article on Xenbase.
Abstract
The African clawed frog, Xenopus laevis, is one of the most widely used model organisms in biological research. However, the most recent anatomical description of X. laevis was produced nearly a century ago. Compared with other anurans, pipid frogs - including X. laevis - exhibit numerous unusual morphological features; thus, anatomical descriptions of more ''typical'' frogs do not detail many aspects of X. laevis skeletal and soft-tissue morphology. The relatively new method of using iodine-based agents to stain soft tissues prior to high-resolution X-ray imaging has several advantages over gross dissection, such as enabling dissection of very small and fragile specimens, and preserving the three-dimensional topology of anatomical structures. Here, we use contrast-enhanced computed tomography to produce a high-resolution three-dimensional digital dissection of a post-metamorphic X. laevis to successfully visualize: skeletal and muscular anatomy; the nervous, respiratory, digestive, excretory and reproductive systems; and the major sense organs. Our digital dissection updates and supplements previous anatomical descriptions of this key model organism, and we present the three-dimensional data as interactive portable document format (PDF) files that are easily accessible and freely available for research and educational purposes. The data presented here hold enormous potential for applications beyond descriptive purposes, particularly for biological researchers using this taxon as a model organism, comparative anatomy and biomechanical modelling.
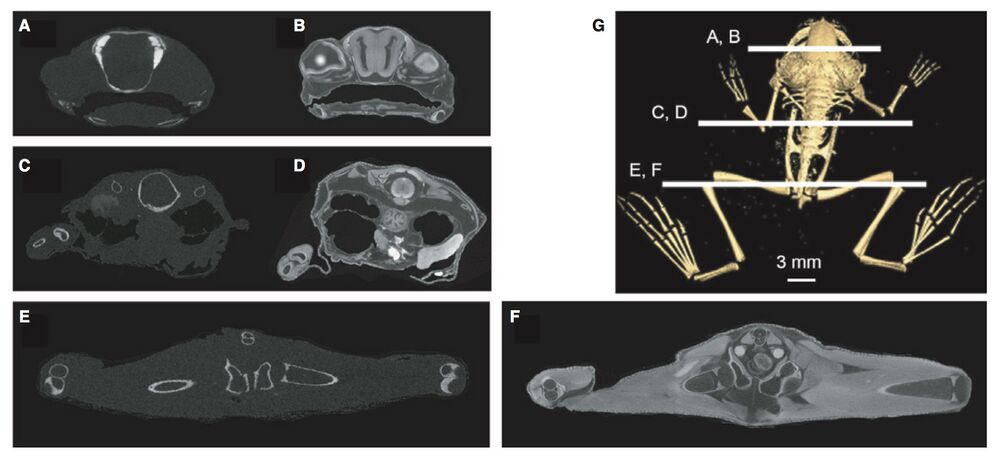
Fig. 1 Coronal/transverse micro-computed tomography (lCT) sections of Xenopus laevis specimen before (A,C,E) and after staining with I2KI (B,D,F). Position of sections through the head (A,B), abdomen (C,D) and pelvis/hind limb (E,F) are shown in (G).
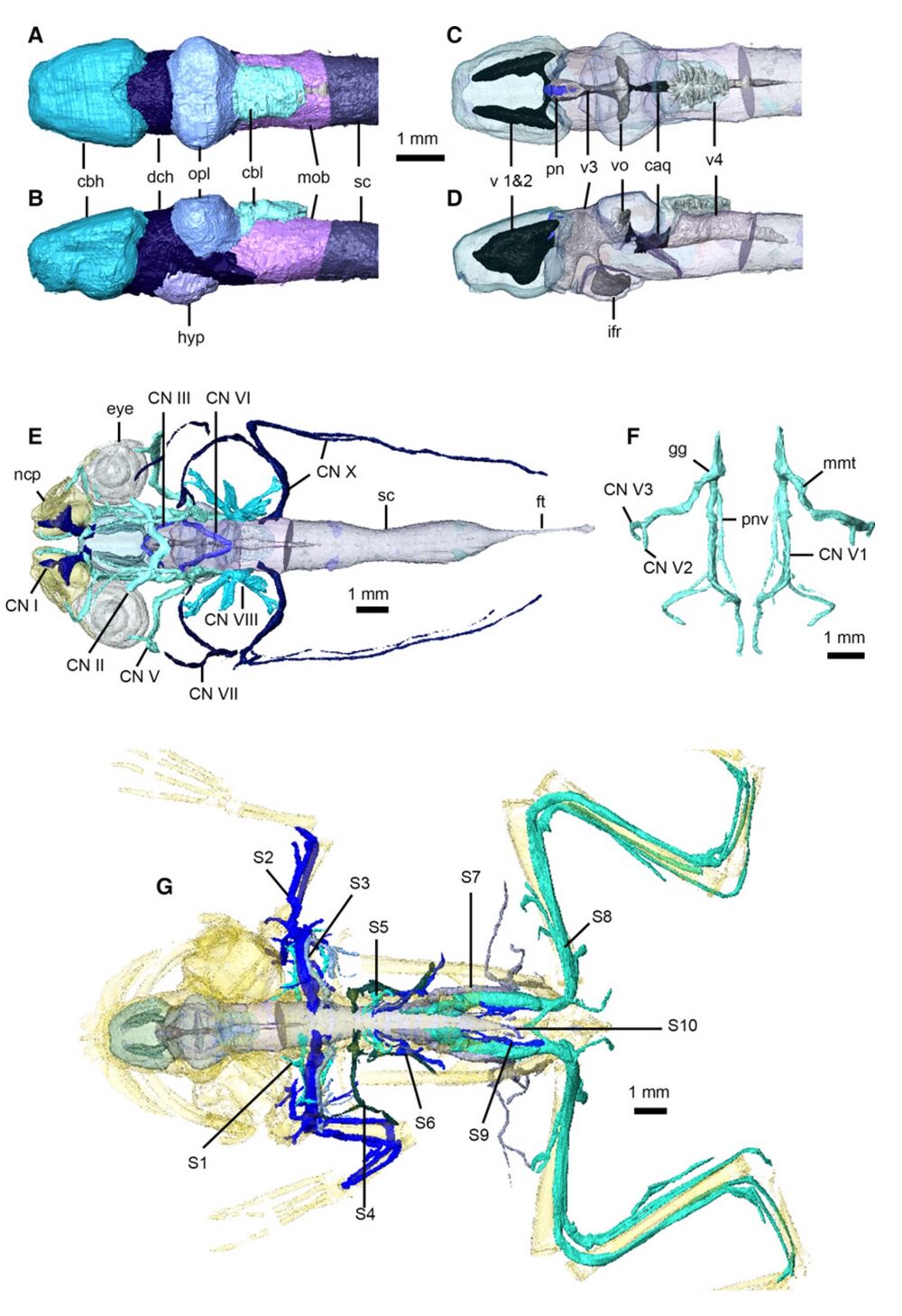
Fig. 11 Nervous system of Xenopus laevis. Brain in dorsal (A,C) and left lateral (B,D) views, with brain transparent in (C) and (D) to illustrate internal ventricles and pineal body. Cranial nerves in dorsal view (E), with eyes, nasal capsules, brain and spinal cord transparent. Left and right trigeminal nerves (F) in dorsal view. Peripheral nervous system in dorsal view (G), with brain, spinal cord and skeleton transparent (cranial nerves not shown). caq, cerebral aqueduct; cbl, cerebellum; cbh, cerebral hemispheres; CN I– X, cranial nerves 1–10; dch, diencephalon; ft, filum terminale; gg, Gasserian ganglion; hyp, hypothalamus; ifr, infundibular recess; mmt, maxilla-mandibular trunk; mob, medulla oblongata; ncp, nasal capsules; opl, optic lobes; pn, pineal body; pnv, palatine nerve; S 1–10, spinal nerves 1–10; sc, spinal cord; v 1 and 2, first and second ventricles; v3, third ventricle; v4, fourth ventricle; vo, optic ventricles.
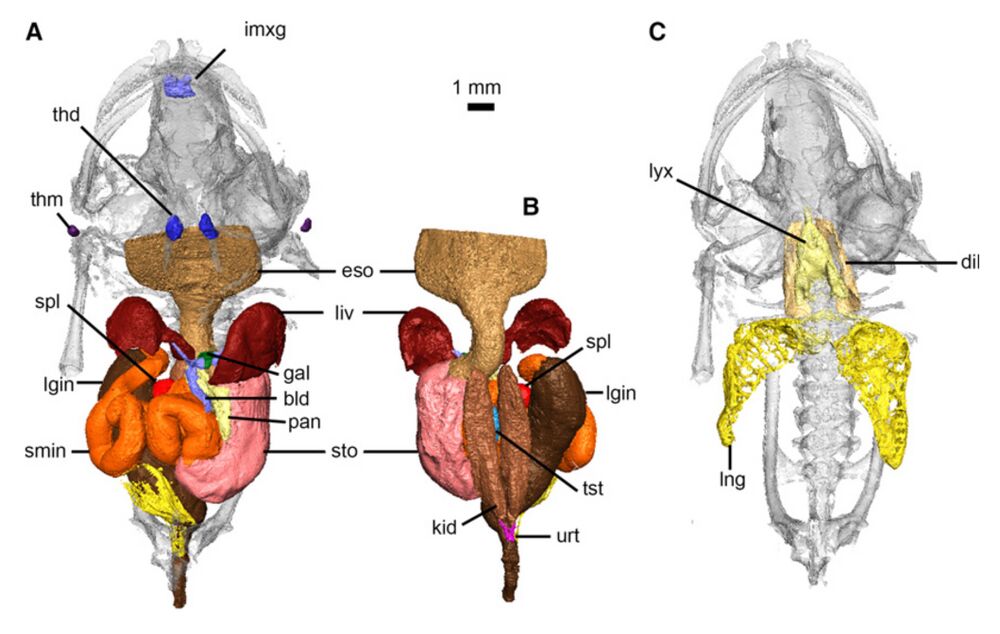
Fig. 12 Digestive, urogenital and respiratory systems of Xenopus laevis. Digestive and urogenital organs in ventral (A) and dorsal (B) views. Respiratory system in ventral view (C). bld, bile and cystic ducts; dil, M. dilator laryngis; eso, esophagus; gal, gallbladder; imxg, intermaxillary glands; kid, kidney; liv, liver; lgin, large intestine and rectum; lng, lung; lyx, larynx; pan, pancreas; smin, small intestine; spl, spleen; sto, stomach; thd, thyroid gland; thm, thymus gland; tst, testes; urt, ureter.
Adapted with permission from the Anatomical Society, Journal of Anatomy: Porro & Richards (2017). Digital dissection of the model organism Xenopus laevis using contrast-enhanced computed tomography. J Anat. August 1, 2017; 231 (2): 169-191. DOI: 10.1111/joa.12625. Copyright 2017.
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
Last Updated: 2017-09-08