XB-IMG-121096
Xenbase Image ID: 121096
|
|
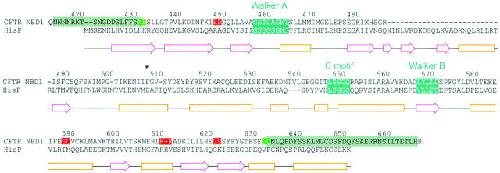
Figure 12. Alignment of CFTR's NBD1 with HisP. PSI-Blast aligned CFTR residues 422–637 with corresponding residues in HisP. Secondary structure elements shown below (β-strands, magenta arrows; α-helices, orange boxes) correspond to those of the solved crystal structure of HisP (Hung et al. 1998). CFTR residue numbers are centered above the corresponding letters. Teal boxes identify the three consensus sequences conserved in most ABC proteins. Red boxes in the CFTR sequence identify nontolerated, green boxes tolerated, cut sites. (Cuts occurred between the two highlighted residues in each case.) Gray boxes identify sections flanking the NBD1 sequence that could be omitted without destroying channel function. *Residue F508. Image published in: Chan KW et al. (2000) © 2000 The Rockefeller University Press. Creative Commons Attribution-NonCommercial-ShareAlike license Larger Image Printer Friendly View |
