Xenopus IgX informs engineering strategies of IgM and IgG hexamers
Xenopus IgX informs engineering strategies of IgM and IgG hexamers
Sci Adv. 2025 Nov 7;11(45):eaea3737. doi: 10.1126/sciadv.aea3737. Epub 2025 Nov 5.
Ruixue Zhang, Chenggong Ji, Shuhan Li, Ningning Li, Ning Gao, Junyu Xiao
Click here to view article at Science Advances.
Click here to view article at Pubmed.
Click here to view article on Xenbase.
Xenopus IgX is a immunoglobulin isotype related to mammalian IgA, and is associated with frog mucosal immunity. While IgA makes dimers, IgX makes hexamers. However, unlike mammalian IgA and IgM in almost all vertebrates, IgX makes multimers without the immunoglobulin J chain (J Immunol. 1985 135(3):1998-2004; Eur J Immunol. 1996 (12):2823-30. doi: 10.1002/eji.1830261205). In this new paper in Science Advances, Xiao and colleagues have shown the basis for the IgX multimerization without the J chain. With these data in hand, they mutated residues in mammalian IgM and IgG to allow those isotypes to form multimers without the J chain. This advance could help in producing large amounts of multimeric immunoglobulins of any isotype in any vertebrate for translational work.
Summary provided by Martin Flajnik, PhD - University of Maryland.
Abstract
Polymeric immunoglobulins are essential components of the immune system in jawed vertebrates. While mammalian immunoglobulin M (IgM) typically forms a pentamer linked by the joining chain (J-chain), Xenopus laevis IgX can assemble into a J-chain-independent polymer. Here, we present the cryo-electron microscopy (cryo-EM) structure of IgX, revealing its hexameric configuration. By incorporating the IgX tailpiece into human IgM, we achieved efficient IgM hexamer formation. Truncating IgM's natural tailpiece to a range of 11 to 16 residues also substantially enhanced hexamerization efficiency. Furthermore, introducing a shortened IgM tailpiece to IgG resulted in effective IgG hexamer formation. We further show that the engineered IgM and IgG hexamers targeting CD20 demonstrated robust complement-dependent cytotoxicity (CDC) against several B lymphoma cells. In addition, the IgG-Fc hexamer functioned as a decoy, attenuating CDC in cell cultures. These findings deepen our understanding of polymeric immunoglobulin evolution and introduce innovative strategies for the development of IgM- and IgG-based biologics.
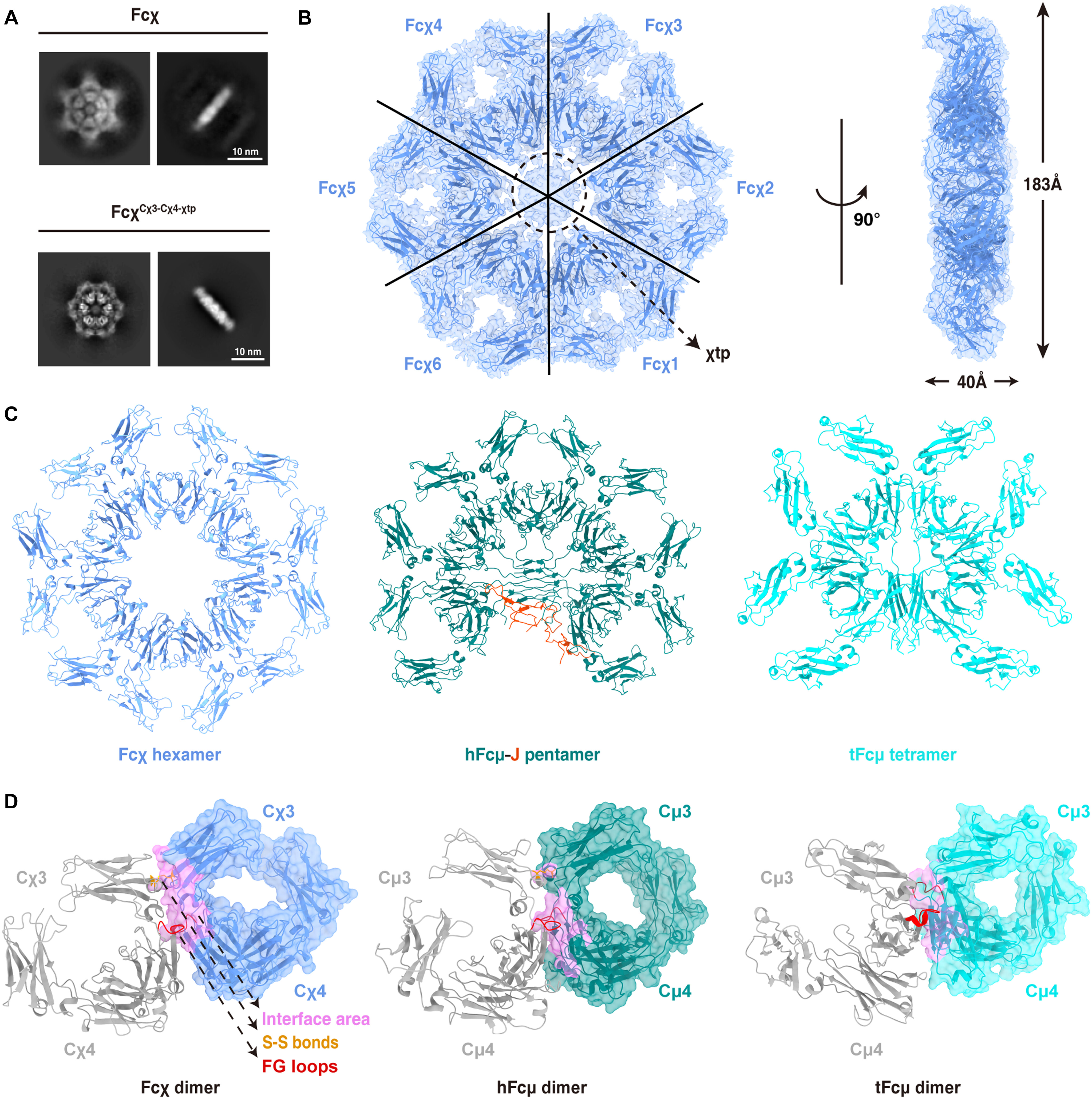
Fig. 1. Xenopus Fcχ can form a stable hexamer.(A) 2D cryo-EM analyses showed that both Fcχ and FcχCχ3-Cχ4-χtp form hexamers. (B) Cryo-EM reconstruction of the FcχCχ3-Cχ4-χtp hexamer at a 3.29-Å resolution, shown in two orientations. The EM density map is superimposed on the structural model. The χtp regions were not clearly resolved because of the blurred densities in this region, as indicated by a dashed circle. (C) Side-by-side comparisons of the Fcχ hexamer (blue), human Fcμ-J pentamer (Fcμ in teal and J-chain in orange), and teleost Fcμ tetramer (cyan). (D) Side-by-side comparisons of the Fc-Fc interactions in the Fcχ hexamer, human Fcμ-J pentamer, and teleost Fcμ tetramer. The interface area, the interchain disulfide bonds, and the FG loops of Cμ4 or Cχ4 domains are highlighted in pink, yellow, and red, respectively.
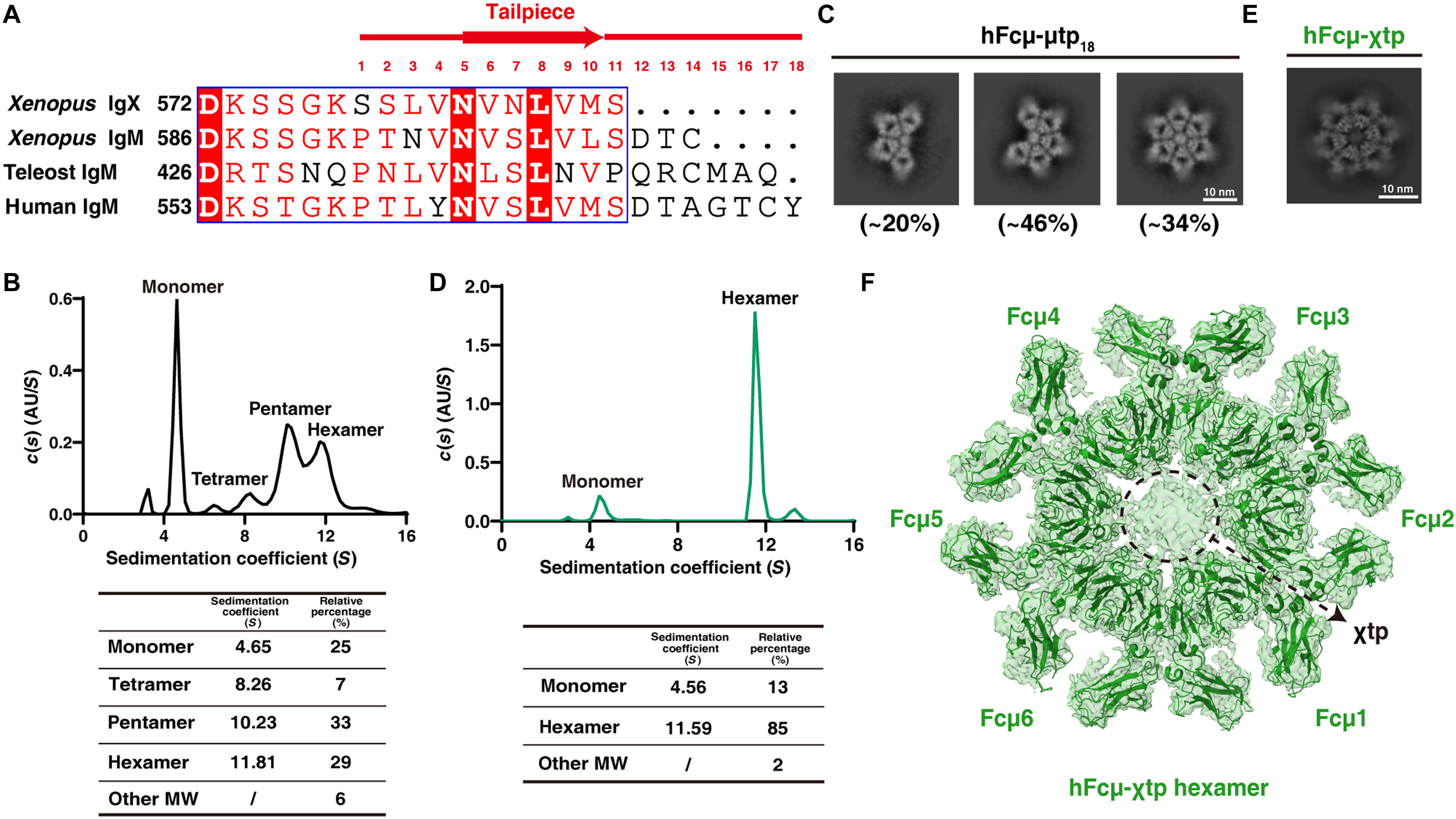
Fig. 2. The Fcμ-χtp chimera forms a hexamer.(A) Sequence alignment of the tailpiece regions of Xenopus IgX and IgM, teleost IgM (from Oncorhynchus mykiss), and human IgM. The tailpiece regions are highlighted. (B) SV-AUC analysis of Fcμ-μtp18 suggested that the monomer, tetramer, pentamer, and hexamer are all present in solution. AU, absorbance units. (C) 2D cryo-EM analyses suggested that the polymers of Fcμ-μtp18 consist of a mixture of tetramer, pentamer, and hexamer. (D) SV-AUC analysis indicated that the Fcμ-χtp polymer is uniformly hexameric in contrast to Fcμ-μtp18. (E) 2D cryo-EM analyses of the Fcμ-χtp chimera. (F) Cryo-EM reconstruction of the Fcμ-χtp hexamer at a 3.29-Å resolution. The EM density map is superimposed on the structural model.
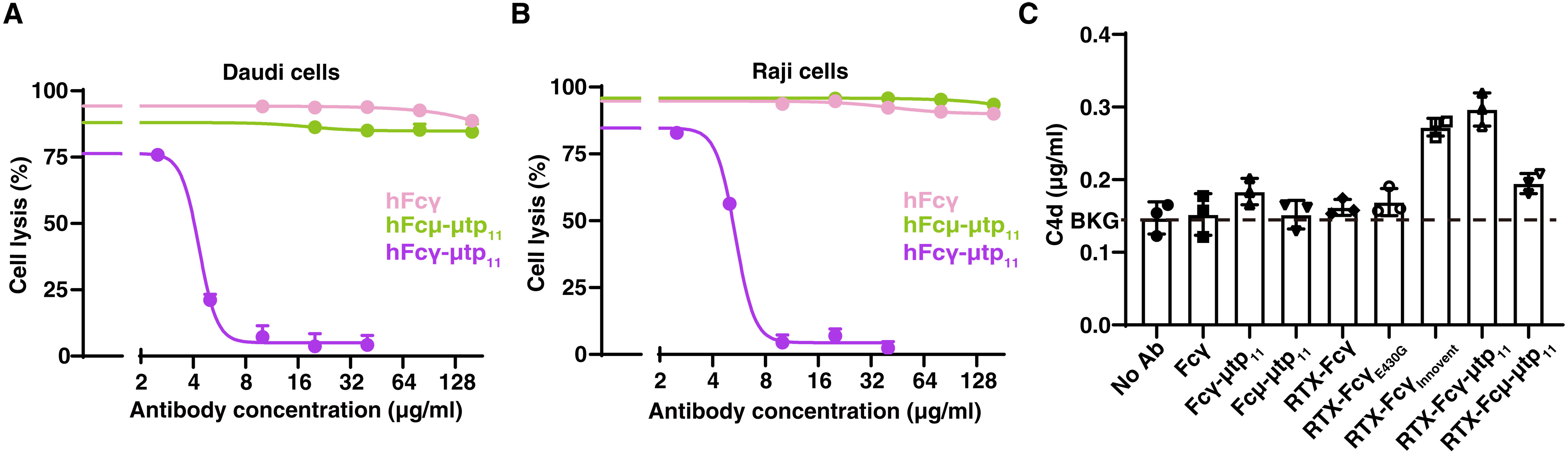
Fig. 6. The engineered IgG-Fc hexamer with μtp11 blocks RTX-IgG–mediated CDC.(A) The IgG-Fc hexamer with μtp11 blocks RTX-IgG–mediated CDC in the Daudi cells. The IgM-Fc hexamers with μtp11 and IgG-Fc monomers exhibit no inhibitory effect. Data were plotted as the means ± SD. n = 3 biological replicates. Source data are provided in the Supplementary Materials. (B) The IgG-Fc hexamer with μtp11 blocks RTX-IgG–mediated CDC in the Raji cells. Data were plotted as the means ± SD. n = 3 biological replicates. (C) Enzyme-linked immunosorbent assay measurement compares C4d generation in human serum. The background (BKG) C4d level in serum is indicated with a dashed line. Ab, antibody. Data were plotted as the means ± SD. n = 3 biological replicates.
Adapted with permission from the American Association for the Advancement of Science on behalf of Zhang et al. Xenopus IgX informs engineering strategies of IgM and IgG hexamers (2025). https://doi.org/10.1126/sciadv.aea3737
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
Last Updated: 2026-02-25